Yes, immortalised cells are safe for cultivated meat. Here’s why:
- Immortalised cells are used because they can multiply indefinitely, ensuring a consistent and scalable supply for production.
- They are not the same as cancer cells. Immortalisation simply means the cells can keep dividing, not that they behave uncontrollably like cancer.
- When consumed, these cells are broken down into basic components (like amino acids) by the digestive system, just like conventional meat.
- Regulatory bodies like the FDA (US) and FSA (UK) rigorously test cultivated meat for genetic stability, contamination, and safety before approval.
- Studies confirm that cultivated meat made from immortalised cells is nutritionally comparable to traditional meat and free from pathogens.
This method reduces the need for repeated animal biopsies, making it a more efficient and ethical production process. Keep reading for a deeper dive into the science and safety measures behind immortalised cells in cultivated meat.
Engineering Cell Lines for Cultured Meat and Sustainable Cellular Agriculture #culturedmeat
sbb-itb-c323ed3
What Are Immortalised Cells?
Immortalised cells are special cell lines that can divide indefinitely, either due to genetic modifications or naturally occurring changes that allow them to bypass cellular senescence. This means they overcome the natural limit on how many times a cell can replicate, known as the Hayflick limit [3].
Normal cells, often referred to as primary cells, have a limited lifespan. Typically, they can only divide 50 to 70 times before they stop replicating and eventually die. Immortalised cells, on the other hand, continue dividing beyond this limit, making them incredibly useful for large-scale manufacturing purposes. This ability to replicate endlessly makes them distinct, as we'll explore in the next section.
"The need to produce immortal, food-relevant cell lines is one of the most pressing challenges of cellular agriculture."
How Immortalised Cells Differ From Other Cell Types
Immortalised cells stand out because they can replicate indefinitely, unlike primary cells, which are limited by the Hayflick limit. Primary cells, once they reach senescence, require fresh biopsies from animals to continue production. In contrast, immortalised cells, once successfully isolated, provide a virtually endless supply of cells, eliminating the need for repeated animal sampling.
Another key difference lies in their adaptability. While embryonic stem cells also have an extensive capacity for replication, immortalised fibroblasts are particularly suited for serum-free suspension cultures. This makes them ideal for use in various bioreactor designs, enabling consistent and efficient production. Since variations between different biopsies are avoided, the resulting product maintains uniformity in taste, texture, and nutritional value [2]. This consistency and scalability make immortalised cells a cornerstone of cultivated meat manufacturing, as discussed below.
Why Cultivated Meat Producers Use Immortalised Cells
One major advantage of immortalised cells is their scalability. These cells can grow as single-cell suspensions in bioreactors without needing a solid surface for attachment. This allows for efficient expansion in industrial-scale systems, such as 300-litre bioreactors.
In December 2022, researchers from Believer Meats demonstrated this scalability in a study published in Nature Food. They successfully grew spontaneously immortalised chicken fibroblasts to a density of 100 million cells per millilitre in a 300-litre bioreactor. Led by Professor Yaakov Nahmias, the team developed stable cell lines (HUN-CF-2 and HUN-CF-4) tailored for serum-free suspension culture. They then transformed these fibroblasts into adipocyte-like cells using soy lecithin, producing a meat alternative that 85% of 120 tasters were willing to adopt as a replacement for traditional meat [2]. This combination of scalability and consistency is vital for cultivated meat production and ties directly to its safety profile, which will be addressed later.
"Immortalisation without genetic modification and high-yield manufacturing are critical for the market realisation of cultured meat."
- L. Pasitka et al., Nature Food [2]
Immortalised cells also align with ethical production practices. By removing the need for repeated animal biopsies, they significantly reduce animal involvement in the process. Additionally, when cells undergo spontaneous immortalisation - gaining the ability to divide indefinitely without genetic modification - it may help avoid some of the regulatory hurdles often associated with GMOs [2].
Common Safety Concerns About Immortalised Cells
The use of immortalised cells in cultivated meat has raised some safety questions among consumers. However, strict monitoring processes and the natural function of human digestion ensure that any theoretical risks associated with these cells do not lead to actual health concerns. One of the primary worries is whether these cells could increase the risk of cancer. To address this, it’s important to clarify that the concept of immortalisation is not synonymous with malignancy.
Do Immortalised Cells Cause Cancer?
The simple answer is no. Immortalised cells differ significantly from cancer cells. Alice Millbank, Co-founder and Chief Scientific Officer at Rooted Research Collective, explains:
"Immortalisation is not the same as malignancy. Instead, it simply means that the cells can keep dividing, whereas cancer involves many additional, uncontrolled behaviours." [1]
Cancer cells demonstrate invasive and unregulated growth within living organisms, but the cells used in cultivated meat are carefully monitored in controlled bioreactor environments. This ensures they remain stable and healthy throughout the production process. Beyond this, the human digestive system plays a crucial role in eliminating any potential risks.
How the Digestive System Processes Immortalised Cells
When consumed, all cells - whether from conventional or cultivated meat - are broken down during digestion into their fundamental components, such as amino acids and nucleotides. This process effectively neutralises any concerns about the cells’ ability to divide indefinitely. Alice Millbank provides further reassurance:
"Even if such cells contain mutations, eating them does not pose a risk; our digestive system breaks down all cells into basic components like amino acids and nucleotides, just as it does with conventional meat." [1]
Since the digestive system completely dismantles these cells, their original characteristics have no bearing on the safety of the cultivated meat vs. conventional meat food safety profile.
Safety Testing and Regulatory Oversight
Before cultivated meat lands on your plate, it must pass through a series of stringent tests and regulatory checks. These measures ensure that the immortalised cells used in its production are safe, stable, and free from contamination.
Testing for Genetic Mutations and Contamination
Genetic stability testing involves advanced techniques like RNA-seq and DNA sequencing to spot mutations. Copy Number Variation (CNV) analysis ensures the genome remains healthy, and Single Nucleotide Variation (SNV) analysis monitors key genes such as TP53 [2]. Additionally, karyotype analysis is used to detect any chromosomal abnormalities within the cell population [2].
Microbial contamination screening is equally thorough, covering bacteria, yeast, fungi, viruses, and mycoplasma. While bacteria and fungi can make culture media visibly cloudy, viruses and mycoplasma are too small to detect visually, requiring specialised molecular tests [5]. Tests like the Limulus Amebocyte Lysate (LAL) or Recombinant factor C (rFC) assays are used to measure harmful bacterial toxins [5]. To confirm the biological origin of the cells and prevent cross-species contamination, species identity is verified through PCR and qRT-PCR [2].
One notable study, published in January 2023, highlighted the work of researchers from Believer Meats (formerly Future Meat Technologies) and The Hebrew University of Jerusalem. The team, including Professor Yaakov Nahmias, demonstrated the spontaneous immortalisation of chicken embryonic fibroblasts. Using RNA-seq, CNV analysis, and SNV analysis, they confirmed genetic stability. Pathogen and pesticide tests were also conducted, comparing the cultivated biomass to commercially available chicken breast to ensure safety [2].
These rigorous testing protocols set the foundation for meeting regulatory standards and associated costs.
UK and International Regulatory Requirements
In the UK, consumer safety is overseen by regulators under the Novel Foods framework, managed by the Food Standards Agency (FSA) and Food Standards Scotland (FSS) [5]. These agencies require detailed safety assessments that address microbiological risks, chemical hazards, nutritional content, allergens, and cell line management before products are approved for the food chain.
Production facilities must follow strict quality protocols, including Good Manufacturing Practice (GMP), Good Cell Culture Practice (GCCP), and Hazard Analysis and Critical Control Point (HACCP) [5]. Real-time monitoring systems, which track variables like pH and dissolved oxygen, help identify early signs of microbial contamination [5]. As Rosario Romero and Emiline Quill from the FSA Research and Evidence team explain:
"The novelty of these food production techniques and the products they generate poses challenges for regulatory bodies like the Food Standards Agency (FSA) and Food Standards Scotland (FSS) in conducting risk assessments based on the best available science." [5]
Globally, similar standards apply. The European Food Safety Agency (EFSA) oversees approvals within the EU, while the US Food and Drug Administration (FDA) handles pre-market safety consultations in the United States [5]. Despite regional differences in regulatory frameworks, the universal goal remains the same: ensuring cultivated meat is safe for human consumption before it becomes available to the public.
Other Cell Types Used in Cultivated Meat
Immortalised cells aren't the only route for producing cultivated meat. In fact, producers have several cell types to choose from, each with its own pros and cons. By examining these alternatives, it becomes clearer why some companies stick with immortalised lines, while others explore different options.
Primary Cells and Embryonic Stem Cells
Primary cells are taken directly from animal tissue via a straightforward biopsy. These cells are naturally inclined to form muscle and fat tissue, making them a logical choice for cultivated meat. But there’s a big drawback: they can only divide a limited number of times before stopping. This means producers would need to regularly return to animal biopsies to keep production going - a logistical nightmare for large-scale operations [6].
At the other end of the spectrum are embryonic stem cells (ESCs). These pluripotent cells have the remarkable ability to become any cell type in the body and can divide indefinitely without needing genetic tweaks. Their rapid growth makes them appealing for scaling up production. However, there’s a challenge. As the Good Food Institute points out:
"Highly potent cells, such as embryonic and induced pluripotent stem cells, have desirable traits such as rapid division and retained immortality. However, their ability to differentiate into any cell type means that extra effort needs to be dedicated to coaxing the cells to differentiate into a single desirable lineage." [6]
Somewhere in between are adult stem cells, such as mesenchymal stem cells (MSCs) and muscle satellite cells. These are more specialised than embryonic cells, making them better suited for turning into muscle and fat tissues. But they divide more slowly and lack the ability to keep dividing indefinitely, which limits cell line longevity and practicality for large-scale, continuous production [6].
These challenges have led to growing interest in other cell sources, particularly naturally immortalised lines.
Naturally Immortalised Cell Lines
Naturally immortalised cell lines present an intriguing option. These cells acquire the ability to divide indefinitely on their own, without requiring genetic modification. This not only simplifies regulatory approval but also appeals to consumers who prefer non-GMO products.
For instance, researchers from the Hebrew University of Jerusalem and Believer Meats have developed spontaneously immortalised chicken fibroblasts. These cells achieved high densities and performed well in sensory evaluations [2].
Other species have seen similar breakthroughs. The Roslin Institute has created a spontaneously immortalised pig pre-adipocyte cell line, nicknamed "FaTTy", which has been successfully cultured for over 64 passages. Meanwhile, DragonB.io offers a porcine spontaneously immortalised adipocyte-derived stem cell line (DB_005), which has surpassed 60 passages [6]. These advancements suggest that naturally immortalised cells could combine the benefits of continuous growth with fewer regulatory concerns compared to genetically modified options.
Weighing the Pros and Cons of Immortalised Cells
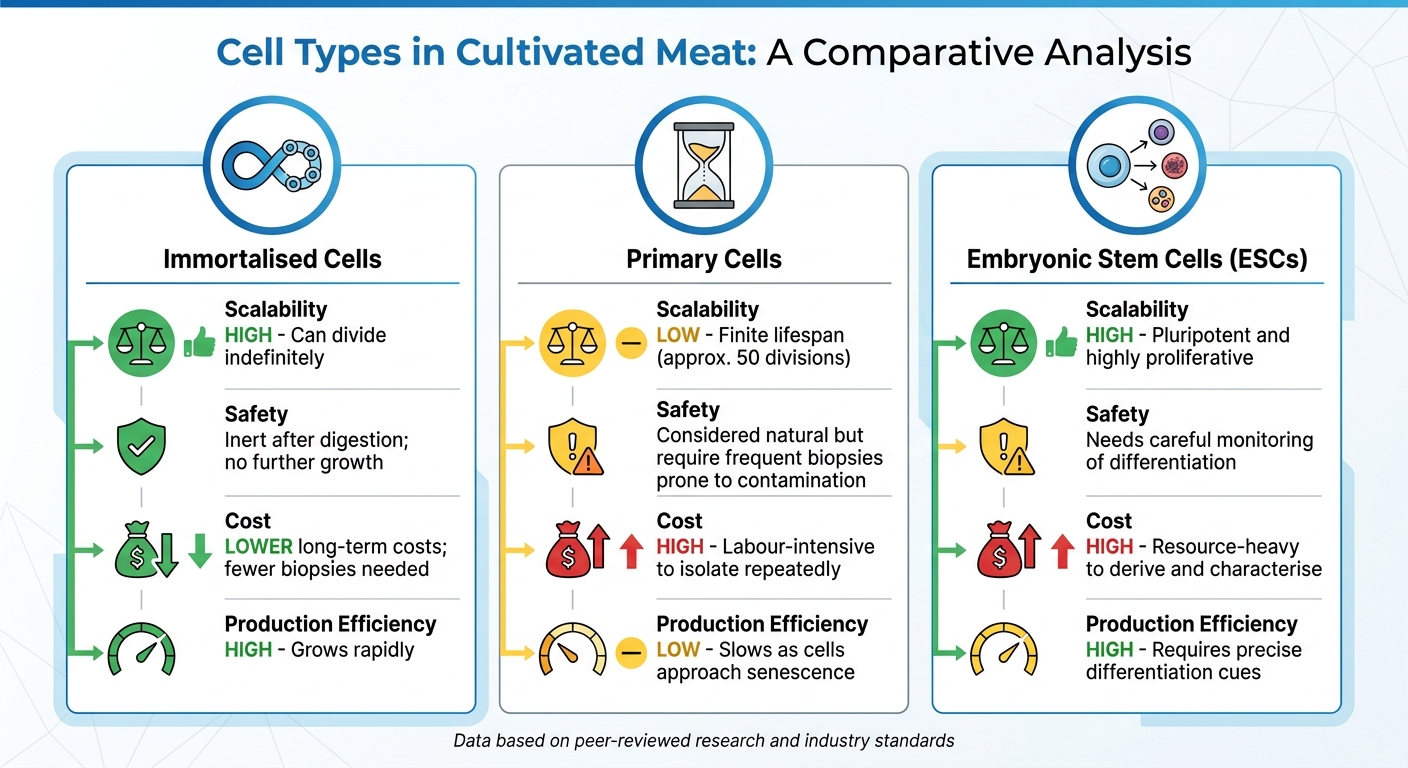
Comparison of Cell Types Used in Cultivated Meat Production
After exploring safety testing and regulatory oversight, it’s time to dig into the upsides and challenges of using immortalised cells. These cells promise scalable production but also raise some safety concerns. The primary advantage? Scalability. Unlike primary cells, which can only divide about 50 times before becoming non-viable, immortalised cells can replicate hundreds of times - or even indefinitely [4]. This makes it possible to produce a continuous supply of meat without the need for repeated animal biopsies.
From a production perspective, immortalised cells are a game-changer. They grow quickly and consistently, which is critical when trying to meet the demand for millions of metric tonnes of meat to feed global populations [4]. As Andrew Stout, Lead Researcher at the Tufts University Center for Cellular Agriculture, puts it:
"Using these new persistent bovine cell lines, studies can be more relevant, literally getting right to the meat of the matter" [4].
While some worry about the potential for these cells to behave unpredictably after consumption, experts assure us that they become inert during processing. David Kaplan, Director of Tufts University Center for Cellular Agriculture, explains:
"While some may question whether it is safe to ingest immortalised cells, in fact, by the time the cells have been harvested, stored, cooked, and digested, there is no viable path to continued growth" [4].
When comparing options, the trade-offs become apparent. Primary cells are often seen as "natural", but they require frequent animal biopsies and can’t scale efficiently. Embryonic stem cells, on the other hand, grow quickly but need precise protocols to ensure they develop into the desired tissue types. Spontaneously immortalised cells, which achieve their longevity naturally without genetic modification, strike a balance by offering continuous growth with fewer regulatory challenges [2].
These benefits come without compromising safety, as rigorous testing and processing ensure the cells are harmless. The table below highlights how immortalised cells compare to primary and embryonic stem cells in key areas.
Comparison: Immortalised Cells vs. Alternative Cell Types
| Factor | Immortalised Cells | Primary Cells | Embryonic Stem Cells (ESCs) |
|---|---|---|---|
| Scalability | High; can divide indefinitely [4] | Low; finite lifespan (approx. 50 divisions) [4] | High; pluripotent and highly proliferative [7][9] |
| Safety | Inert after digestion; no further growth [4] | Considered natural but require frequent biopsies prone to contamination [4][8] | Needs careful monitoring of differentiation [7] |
| Cost | Lower long-term costs; fewer biopsies needed [4] | High; labour-intensive to isolate repeatedly [4] | High; resource-heavy to derive and characterise [7] |
| Production Efficiency | High; grows rapidly [4] | Low; slows as cells approach senescence [4] | High; requires precise differentiation cues [7] |
This breakdown shows why immortalised cells are becoming the preferred choice for large-scale production. Their ability to grow indefinitely, combined with lower costs and high efficiency, makes them a practical solution for the cultured meat industry.
Conclusion: The Safety of Immortalised Cells in Cultivated Meat
The evidence is clear: immortalised cells used in cultivated meat are safe for human consumption. Extensive testing has shown these cells are genetically stable, free from pathogens, and incapable of further division after processing, ensuring their safety [2].
In January 2023, a study conducted by Believer Meats and The Hebrew University of Jerusalem, under the guidance of Professor Yaakov Nahmias, demonstrated that spontaneously immortalised chicken fibroblasts maintain genetic stability even during long-term cultivation. The study also revealed that these cells produce biomass free from pathogens and pesticides, with a nutritional profile comparable to conventional chicken [2]. Impressively, the cultivated chicken scored 4.5 out of 5.0 in sensory tests, and 85% of participants expressed a strong likelihood of replacing traditional meat with cultivated alternatives [2].
Regulatory measures further ensure safety by monitoring critical tumour-suppressor genes like TP53 and PTEN, while also confirming nutritional equivalence. The natural process of immortalisation in these cells addresses concerns about genetic modification [2].
To learn more about the safety measures and advancements shaping cultivated meat, check out educational resources or join the waitlist at Cultivated Meat Shop.
FAQs
How are immortalised cells created?
Immortalised cells are specially developed to keep dividing indefinitely, a key requirement for producing cultivated meat. This can happen in two ways: spontaneous immortalisation, where cells naturally gain the ability to divide endlessly, or through genetic modification, where specific genes are altered to achieve the same result. Both approaches are thoroughly tested and reviewed to meet strict safety and regulatory standards, ensuring they are fit for use in food production.
What safety checks happen before cultivated meat is approved in the UK?
Before cultivated meat can be approved in the UK, it must pass extensive safety checks. The production process takes place in sterile environments equipped with HEPA filters and bioreactors to minimise contamination risks. Advanced testing methods, such as DNA staining, PCR detection, and genetic sequencing, are used to identify any potential contaminants. Additionally, the process must comply with the rigorous safety standards established by regulatory bodies like the FSA. These steps ensure the meat is safe and meets high-quality standards before it becomes available to consumers.
Will cultivated meat made from immortalised cells affect allergies or digestion?
Cultivated meat, created using immortalised cells, is developed under rigorous safety protocols. Studies indicate that it carries no extra risks for allergies or digestion when compared to traditional meat. Advanced purification techniques and strict regulatory measures are employed to manage residual proteins and minimise any potential cross-contact, ensuring the product remains safe for consumption.













